 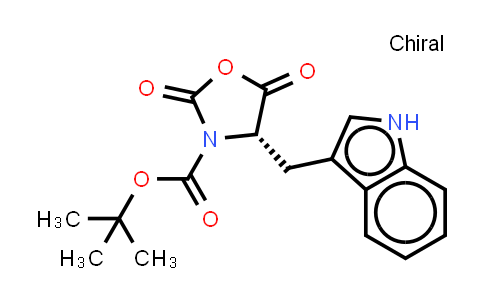
Then, you're stuck chipping away at it with a metal spatula. That is, unless you keep it on your bench, in which case it doesn't go (totally) bad, but it will probably melt a bit, depending on the weather. It comes as beautiful melty clear crystals, kind of like soft-cornered rock candy with very small crystals. Boc homolog (mouse), BOC and IDBG-50192 and ENSG0000014483, protein binding, Plasma membranes, Boc and IDBG-162156 and ENSMUSG00000022606, BOC and IDBG-632359 and ENSBTAG00000013918. Additionally, it comes in great big bottles - it's not full of the stuff you need a little headspace for carbon dioxide gas. Such is chemistry - it's the best we've got! At least it's fun to say boc (pronounced like "bock").īoc anhydride tends to decompose a little on storage (and it melts around room temperature - it's a pain chipping away at a slab of melted-refrozen stuff). He doesn't go on to explain it further, but the analogy is apt - they're not bolts, and are prone to fall apart. As Derek Lowe mentions in his post, it's kind of like a piece of masking tape. Protecting groups are finicky things and don't always work as you expect. We offer qualified product 1-Octenylsuccinic anhydride (), please inquire us for. Using Boc will convert an amine into a carbamide that won't react with (as many) things, allowing you to work unfettered (or at least less fettered) on other chunks of whatever molecule you're interested in. BOC Sciences is the world-leading provider of special chemicals. The latter two are both gases, which gives about as easy a cleanup as you could hope for (that's the problem with these protecting groups you have to get rid of them somehow). Using it on your amine will yield a "boc protected amine." Upon acid deprotection, it yields the parent amine, carbon dioxide, and t-butylene. Protecting groups are ubiquitous in organic synthesis, and another one you see all the time is Boc, which is used to protect amines:īoc 2O, or "boc anhydride," is one of my favorites. The formation of Boc-protected amines and amino acids is conducted under either aqueous or anhydrous conditions, by reaction with a base and the anhydride Boc 2 O. P501 Dispose of contents/container to an appropriate treatment and disposal facility in accordance with applicable laws and regulations, and product characteristics at time of disposal.One of my best friends in undergrad, upon learning about the use of acetal protecting groups for carbonyls, exclaimed, "they're like ketone condoms!" Indeed. P403+P233 Store in a well-ventilated place. P319 Get medical help if you feel unwell. P304+P340 IF INHALED: Remove person to fresh air and keep comfortable for breathing. Remove contact lenses, if present and easy to do. P305+P351+P338 IF IN EYES: Rinse cautiously with water for several minutes. P332+P317 If skin irritation occurs: Get medical help. P362+P364 Take off contaminated clothing and wash it before reuse. P302+P352 IF ON SKIN: Wash with plenty of water/. P301+P317 IF SWALLOWED: Get medical help. Boric anhydride B2O3 - PubChem Apologies, we are having some trouble retrieving data from our servers. Di-tert-butyl dicarbonate, BOC 2 O (also known as di-tert-butyl pyrocarbonate or BOC anhydride), triethylamine (TEA), and tetrahydrofuran (THF) are commonly used to add the BOC group. 
P271 Use only outdoors or in a well-ventilated area. The BOC (tert-butoxycarbonyl) group is used to protect amines in synthetic reactions. P261 Avoid breathing dust/fume/gas/mist/vapours/spray. P280 Wear protective gloves/protective clothing/eye protection/face protection/hearing protection/. P270 Do not eat, drink or smoke when using this product. L-Tyrosine,N-O-(phenylmethyl)- Alanine,3-N-carboxy-,N-tert-butyl ester,L- Alanine,3-N-carboxy-,N-tert-butyl ester L-Alanine,3-N-carboxy-,N-tert-butyl ester N-O-(phenylmethyl)-L-tyrosine N-(tert-Butyloxycarbonyl)-O-benzyl-L-tyrosine O-Benzyl-N-tert-butoxycarbonyl-L-tyrosine N-(tert-Butoxycarbonyl)-O-benzyl-L-tyrosine (tert-Butoxycarbonyl)-O-benzyltyrosine N-(tert-Butoxycarbonyl)-O4-benzyl-L-tyrosine N-(tert-Butoxycarbonyl)-O-benzyltyrosine NSC 164044 (2S)-3-2-propanoic acid (S)-3-(4-Benzyloxyphenyl)-2-(tert-butoxycarbonylamino)propionic acid (2S)-2-3-(4-phenylmethoxyphenyl)propanoic acid (S)-3-(4-(Benzyloxy)phenyl)-2-((tert-butoxycarbonyl)amino)propanoic acid (2S)-3-(4-Benzyloxyphenyl)-2-(tert-butoxycarbonylamino)propanoic acid 11 12 5 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
